Why is it so hard to find a vaccine for HIV?
The first large HIV vaccine trial in seven years is set to start in South Africa - many others before have failed.
When HIV was found to be the cause AIDS in 1984, the US department of Health hoped to have a vaccine ready within the next couple of years.
More than three decades later, a safe and effective preventive vaccine has yet to be licensed and more than 250 clinical trials have failed. A large study set to begin in South Africa on the eve of World AIDS day, held on 1 December 2016, is proving to be scientists' best hope in seven years.
The clinical trial, known as HVTN 702, involves a new version of the only HIV vaccine candidate ever shown to provide some kind of protection against the virus – the one tested in the RV144 clinical trial in Thailand, led by the US Military HIV Research Program.
The vaccine candidate appeared to be 31.2% effective at preventing HIV infection over a 3.5 year testing period.
Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious diseases, one of the trial's funders, told IBTimes UK: "A vaccine with a 31.2% efficacy is not something you want to widely distribute, but it is enough to encourage you to try and expand upon it.
"That is exactly what this trial is about, it uses a similar platform– a canarypox-vector based vaccine with multiple gene inserts for viral proteins, followed by multiple boosts of the protein itself, together with an adjuvant – to reach greater efficacy on the longer-term. If we reach a 50 or 60% efficacy, I would consider the trial quite successful."
Expectations are high, as such a success would mean renewed hopes to curb the number of new infections globally. There were 2.1 million new cases reported in 2015, bringing the total of people living with HIV around the globe to 36.7 million.
IBTimes UK takes a look at why finding a vaccine for HIV is so complicated and how the HVTN 702 trial builds on previous efforts.
Read more about HVTN 702: First large-scale HIV vaccine trial in seven years to start in South Africa
Do better than 'nature'
HIV is very different from all the other viruses for which a vaccine has successfully been developed. Diseases like polio or small pox have different degrees of morbidity and mortality, but in most people who become infected, the viruses elicit an effective immune response in the body that ultimately clears the virus and protects the person from reinfection with the same virus. Vaccines for the two diseases mimic this protective response.
"Back in the day when there was still small pox, mortality was 15 to 20%. This means that 80 to 85% of people survived and did well in the long term," said Fauci. "With polio, more than 90% of people ultimately do well. So the body proves that it is capable of mounting an effective immune response.
"Thus, natural infection has provided a 'proof of concept' indicating that it is feasible and likely that you will be able to reproduce this natural immune response with a vaccine, based on this successful experiment of nature."
The problem with HIV is that the body does not easily mount a good immune response against HIV. Among people who are infected, virtually no one goes on to clear the virus or keep it at low level – with the exception of elite controllers who represent less than a fraction of 1% of all HIV infections. This means that to find an effective HIV vaccine, scientists have to do 'better than nature'.
"When researchers start to think about developing HIV vaccines, they face the problem of having to do better than natural infection. The body does not make a good immune response, and so we have to figure out a way to push the body to create something that it does not readily do naturally. This is why it has been so hard to find a vaccine", Fauci said.
Blocking the infection at the 'get-go'
HIV is a retrovirus, which means it is composed of RNA. An enzyme gives it the unique property of transcribing its RNA into DNA after entering a host cell. This retroviral DNA then integrates into the chromosomal DNA of the host cell. This means that typical approaches to deal with the the virus don't necessarily work.
Robert Gallo, the Director of the Institute of Human Virology at the University of Maryland School of Medicine, who co-discovered that HIV was the cause of AIDS, told IBTimes UK: "We can't sufficiently learn from classical virology, because retroviruses are completely different. Retroviruses integrate. In one day the genes of the virus are part of you, of your target cells and their progeny."
"Within a few days, these cells are making variants of the virus and it's too late for the immune system to react. It means that we have to have a broadly reactive immune response, capable of blocking virus entry at the get-go".
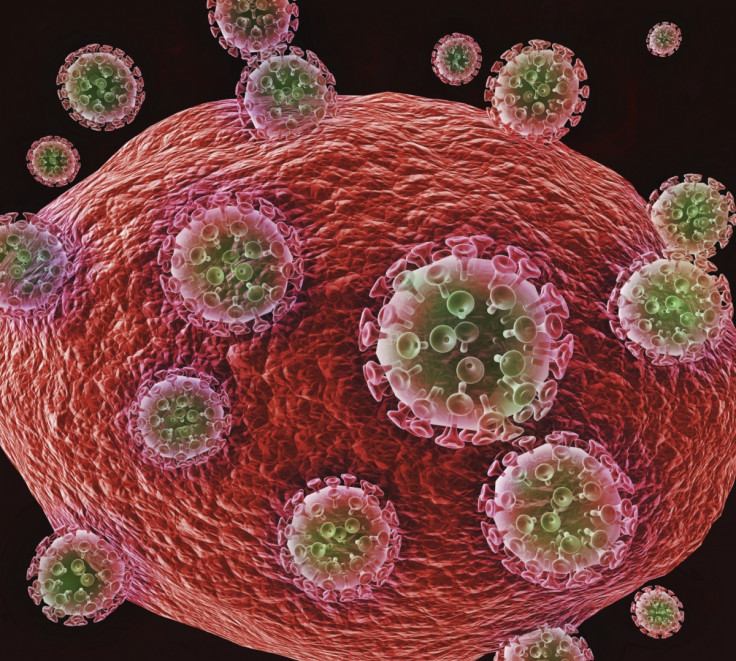
An effective preventive vaccine will have to constantly maintain this broad immune response, as there is no time for a recall of antibodies when a person is infected. This is an important challenge of vaccine development.
The way forward
The question now is whether the HVTN 702 trial will succeed in addressing these different challenges and whether it will do better than the RV144 trial in Thailand in 2009.
"Even if a vaccine candidate shows only modest protection and efficacy, it still can allow you to identify a correlate of immunity and you can then design new vaccines to amplify and improve on that correlate. This is the approach we have chosen here in this trial, learning from the RV144 trial and improving the vaccine regimen to provide greater and more sustained protection", Fauci said.
He adds that some of the other interesting approaches tested at present focus on inducing broadly neutralising antibodies in the vaccine recipient.

Gallo and his team are developing a vaccine candidate to stop the virus before it even enters the cells. "We must block infection from the very beginning, before it gets into the cell. This means we need a vaccine that attacks HIV's protein envelope gp120 with antibodies", Gallo said.
For HIV to infect immune cells, gp120 must first bind to CD4 receptors on the surface of these cells and then transition to bind to another receptor known as CCR5 on immune cells. The vaccine Gallo works on aims at eliciting broadly neutralising antibodies that can link to HIV's gp120 envelope during its transition state, stopping it from binding to CCR5 and thus blocking the infection.
Some scientists have argued in the past that more basic research will be needed before a vaccine can be found. But vaccine trials are in themselves a good way to learn about HIV. Even if the HVTN 702 study or Gallo's research are not successful, their work will surely provide scientists with valuable additional knowledge about the virus.
Phindile Happiness Dlamini's story
Nearly 5,400 healthy, sexually active South African men and women aged between 18 to 35-years-old are enrolled in the HVTN 702 trial, with results expected in late 2020.
Phindile Happiness Dlamini is one of them. An 18-year-old student of Operations and Quality management at Durban University of Technology, she comes from KwaMashu, a working class neighbourhood north of Durban. HIV rates are not higher there than in the rest of country, but stigma against people living with HIV is still significant.
"I think that youth are not taught about HIV prevention at an early age. I was introduced to the trial by my older sister, who was a participant in a previous trial at the research clinic. I attended a pre-screening workshop, where I was informed about the vaccine study. I learnt about how the vaccine is made and, about HIV in general. I was fascinated, and became motivated to participate", Dlamini told IBTimes UK.
In South Africa, young people often lack basic knowledge about HIV and other sexually transmitted diseases because of a limited access to reproductive health services. "For 18-year-old females, it's considered shameful to access reproductive services. The nurses in the local government clinic are sometimes not approachable. Adults still have the belief that teenagers should not be having sex", said Dlamini.
Thus, the trial is also an opportunity to expose thousands of young South Africans to information about HIV that they might not have had access to before.
"I just hope that the vaccine results are good enough, that it helps future generations. I feel that our communities need a vaccine as it would mean a chance to live in an HIV-free world", Dlamini said.
© Copyright IBTimes 2025. All rights reserved.