3D Printer Creates Battery as Small as Grain of Sand to Power Tiny Robots

Scientists have developed a battery the size of a grain of sand with a 3D printer to power miniscule devices.
The researchers say that over recent years, many products used in communication and medicine have had to be put to one side as there was no battery small enough to power them.
Products developed include medical implants, tiny cameras, microphones that fit on glasses and flying insect-like robots.
The batteries that power them, however, were often as big as the device itself, defeating their purpose.
In order to develop the microbatteries, the team from Harvard University and the University of Illinois used a 3D printer to interlace stacks of battery electrodes that measure less than the width of a human hair.
Previously, tiny batteries were made by depositing films of solid material to build electrodes but because of their design, they did not have enough energy to power micro devices.
The team realised they could generate more energy if they could stack tightly interlaced electrodes.
Jennifer Lewis, senior author of the study, said: "Not only did we demonstrate for the first time that we can 3D-print a battery, we demonstrated it in the most rigorous way."
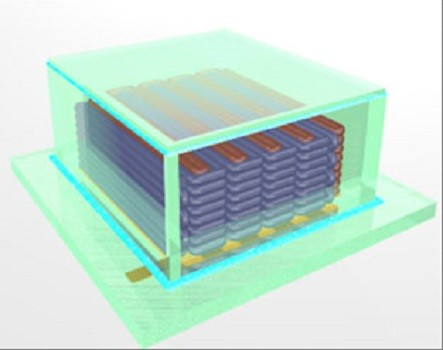
The 3D printer created the microbatteries from computer drawings. The printer deposited layers of material to build the physical object from the ground up.
"Unlike the ink in an office inkjet printer, which comes out as droplets of liquid that wet the page, the inks developed for extrusion-based 3D printing must fulfil two difficult requirements," a statement from Harvard said.
"They must exit fine nozzles like toothpaste from a tube, and they must immediately harden into their final form.
"In this case, the inks also had to function as electrochemically active materials to create working anodes and cathodes, and they had to harden into layers that are as narrow as those produced by thin-film manufacturing methods."
The researchers made two inks which were then deposited onto gold combs that created the stack. The electrodes were then packaged into a container and filled with an electrolyte solution, giving the end battery.
Co-author Shen Dillon said: "The electrochemical performance is comparable to commercial batteries in terms of charge and discharge rate, cycle life and energy densities. We're just able to achieve this on a much smaller scale."
Donald Ingber, founding director of the Wyss Institute at Harvard, said the battery opens up a realm of possibilities for miniaturising all devices: "It's tremendously exciting," he said.
© Copyright IBTimes 2025. All rights reserved.






















