3D Printed Bionic Eye Prototype Successfully Tested to Enhance Vision in Sight-Loss Patients

A bionic eye generated using a 3D printer has been successfully tested on individuals suffering from both partial and total vision loss.
Researchers at the Bionics Institute in Melbourne, Australia, have been developing bionic eye technology that can be used to treat retinitis pigmentosa and age-related macular degeneration.
In order to more efficiently produce prototypes, senior research fellow at the Bionics Institute, Chris Williams, worked with 3D printing company evok3d to utilise a ProJet 1200 3D printer in the process.
"We can now get a prototype out in 4 hours using the ProJet 1200," Williams told 3D Print. "Before 3D printing it would take us weeks or months. We found it takes 20 iterations to reach an upgrade; in terms of going through iterations, the machine justified itself in the first week."
The bionic vision system still remains in its testing stages but already several individuals have had the device inserted and experienced enhanced vision through the first clinical trials.
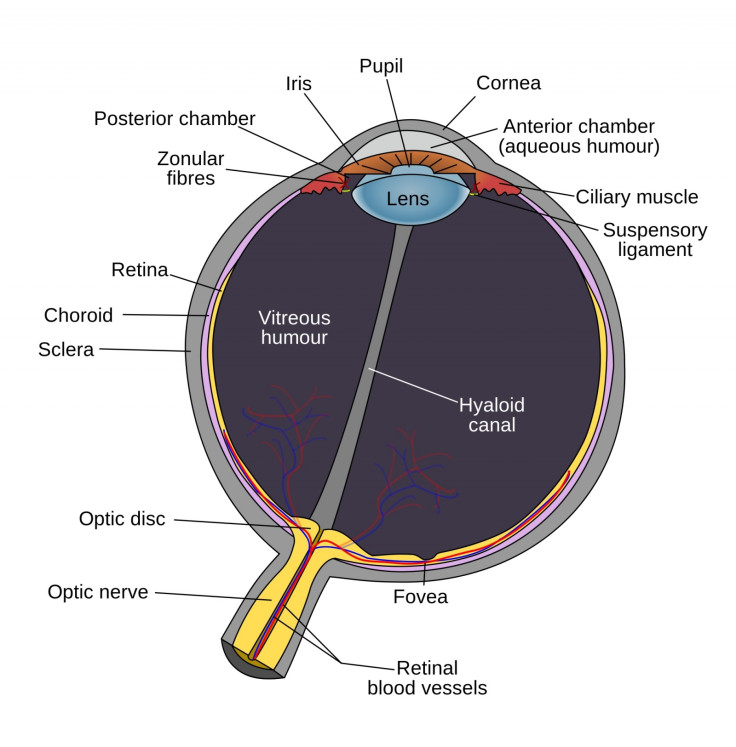
In order for it to have an effect on sufferers from pigmentosa and macular degeneration - which together account for more than 10% of visual impairment causes - the individual needs to still have a healthy optic nerve and visual cortex, as well as some remaining retinal ganglion cells.
Radio signals are transmitted from a camera integrated into the bionic eye and received by a microchip at the back of the eye by the optic nerve. Electrical impulses are then sent out from the chip to stimulate retinal cells to send an image through the optic nerve to the brain.
Currently the technology does not restore full sight but can allow blind individuals to see "blob-like" shapes and therefore navigate new areas unassisted.
Researchers at the University of New South Wales in Australia have previously predicted that a fully-working bionic eye will be commercially available by 2020.
"It was quite promising, their vision was optimised," Williams said. "Obviously they want better vision and fully wireless power, but the eye surgeons were pleased with the process and that's a platform for future trials."
© Copyright IBTimes 2025. All rights reserved.






















