Toxic Peruvian tarantula venom has potential to be used as a strong painkiller for chronic pain

Venom from tarantulas could be used as a treatment for chronic pain, say researchers from the University of Queensland. The scientists say that they can use a single peptide from within the venom to inhibit the function of a pain receptor in the brain.
The peptide, ProTx-II, is found within the venom of the Peruvian green velvet tarantula. The researchers say that ProTx-II interacts with the pain receptor known as Nav 1.7. The research was discussed at the Biophysical Society's 60<sup>th Annual Meeting.
Sónia Henriques, researcher working on the study said: "Our group is specifically interested in understanding the mode of action of this toxin to gain information that can guide us in the design and optimization of novel pain therapeutics."
The research team knew before the study started that the peptide could work as a painkiller in theory, but they did not know exactly which pain receptor they should target.
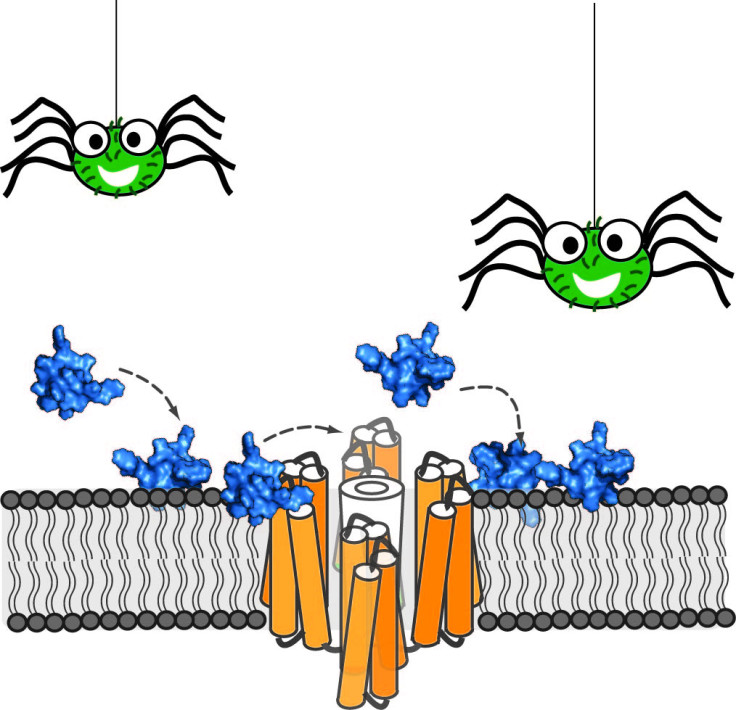
Therefore, they used nuclear magnetic resonance (NMR) spectroscopy on the peptide – enabling them to look at its 3-D structure to isolate which type of receptor it could be used against.
"Our results show that the cell membrane plays an important role in the ability of ProTx-II to inhibit the pain receptor," said Henriques. "In particular, the neuronal cell membranes attract the peptide to the neurons, increase its concentration close to the pain receptors, and lock the peptide in the right orientation to maximize its interaction with the target."
This is the first study which describes the properties of ProTx-II, and its ability to bind to the Nav 1.7 pain receptor.
More precisely, ""It binds to the pain receptor located within the membrane of neuronal cells," said Henriques.
The scientists are now researching new toxins which would be more effective than the ProTx-II peptide. Whilst still suggesting the peptide would be an effective painkiller, they believe they can find another one which has a much bigger affinity to the receptor – imagine the receptor is a magnet; the scientists are trying to find a metal (peptide) with a bigger pull towards that magnet.
Henriques added: "Our work creates an opportunity to explore the importance of the cell membrane in the activity of peptide toxins that target other voltage-gated ion channels involved in important disorders."
© Copyright IBTimes 2025. All rights reserved.




















