Wireless Energy Transfer Breakthrough Heralds New Ways of Treating Diseases

Stanford researchers have invented a method of transferring power to tiny electronic devices implanted in the body, promising innovative new ways of treating diseases.
The breakthrough could pave the way for a new generation of electroceutical devices, which use electrical stimulation rather than traditional medicines to affect and modify the functions of the body.
The technology allows doctors to treat diseases using electronics rather than drugs. Using the body's bioelectric signals rather than its chemistry, electroceuticals are capable of treating a wide range of diseases and conditions, including asthma, diabetes, depression, heart failure and chronic pain.
The discovery by Stanford electrical engineer Ada Poon and her team of researchers is published in the Proceedings of the National Academy of Sciences journal.
In current medical implants, the battery is the largest part of the device and can be difficult to recharge or can require surgery to be replaced.
"We were inspired by the disparity between the size of the microchips that are made today and the bulkiness of medical devices," John Ho, an electrical engineer involved in the research, told IBTimes UK.
"The past few decades have brought about dramatic miniaturisation of electronics, yet because of the lack of a small power source, electronic implants have remained relatively large, at least several centimetres in size."
The device built by the team, which works as a pacemaker despite its miniature size, can be recharged wirelessly with an external power source about the size of a credit card.
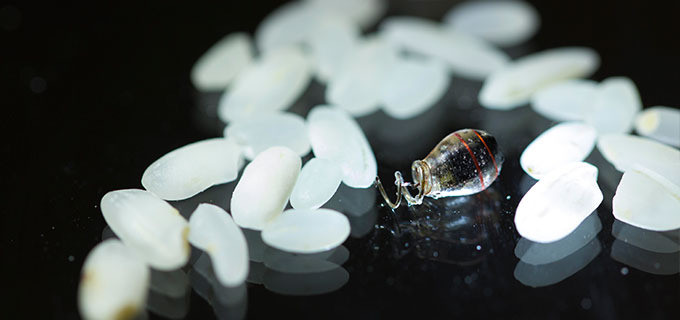
To eliminate the need for bulky batteries, flat coils were used to wirelessly transfer power to an electroceutical the size of a grain of rice implanted deep inside the body
The miniature devices can be used in almost all parts of the body - including the heart and brain - in order to stimulate nerves, implant micro-sensors and deliver drugs to specific areas, while lowering risk, decreasing side-effects and delivering greater benefits than traditional medicines.
"Using the powering method, new generations of miniaturised sensors and stimulators could act as electronic 'medicines' that are more effective than drugs for some diseases," Ho said.
The wireless charging system has already been tested in a pig and used to power a pacemaker in a rabbit, with human trials currently being prepared.
"Clinical trials will require at least a year for approval and further system development, while commercial products that use the powering method may take five to ten years," Ho said.
© Copyright IBTimes 2025. All rights reserved.