HIV 'cured' in monkeys with antibody therapy leading to undectable levels of virus
An antibody known as anti-alpha4/beta7 has led monkeys to control infection even after two years.
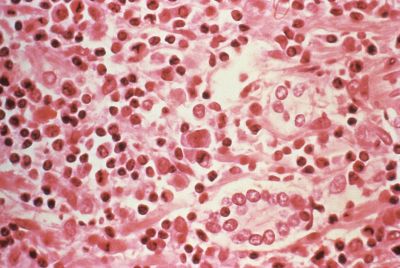
Scientists have shown that achieving a sustained control of an SIV infection in macaques - the simian equivalent of an HIV infection in humans - is possible. Giving the animals an antibody during and after treatment with antiretroviral therapy (ART) appears to restore the infected animals' immune system and to block the re-emergence of the virus once ART is stopped.
ART has been one of the most important advances in the fight against HIV. The treatment, which consists of a combination of antiretroviral drugs, works by suppressing the HIV virus and thus stopping the progression of the disease. ART is credited with having dramatically reduced the number of AIDS-related deaths – it is believed that with prevention, ART has saved 7.8 million lives over the last 15 years.
However, antiretroviral therapy is a life-long treatment with a number of important side effects such as gut damage, digestive problems, nausea or anaemia. This means that there are issues with treatment compliance and that patients' quality of life can be reduced.
More importantly, ART does not 'cure', as the virus persists in the body, in a latent form, inside hard-to-eradicate reservoirs. If ART is discontinued, the viral loads rapidly rebounds and the number of CD4+ T immune cells decreases.
In this research, published in the journal Science, the researchers therefore sought a new method to reduce patient dependence on ART. The antibody they have tested in non-human primates, called anti-alpha4/beta7, appears to do just that – it leads to a sustained control of the virus without the need to take antiretroviral drugs on the long-term.
What the discovery means
The researchers have worked with macaques infected with SIV. Nine weeks after infection and four weeks after starting ART, they started giving monkeys anti-alpha4/beta7 infusions once every two weeks for a total of 32 weeks. They compared them with a control group of seven macaques.
When antiretroviral drugs were discontinued, SIV came back in full force in the seven control animals, even though it had barely been detectable before. Six of the eight alpha4/beta7 treated animals also showed some rebound of viral levels, but they controlled it within four weeks. The other two monkeys treated with the antibody never even showed a rebound of the virus.

Two years after this animal trial was conducted, the monkeys treated with the antibody still controlled the infection. They achieved a 'sustained infection control' – the virus is still present in their bodies, but below the limit of detectability in their blood. The macaques' immune system also shows signs of restoration, as the researchers observed a gradual restoration of CD4+ T cells, the main target cells for the virus, and other immune cells.
"This is an important discovery. I believe that what we have done here is come up with a model for an alternative to ART. If it can be translated in humans, it will reduce the cost of ART and improve patients' quality of life", lead author Aftab A. Ansari from Emory University school of Medicine, told IBTimes UK.
How the antibody works
The anti-alpha4/beta7 antibody works by stopping susceptible CD4+ T cells from circulating into the gut. It is thought this may protect them from being destroyed by the virus during an infection.
The primary target of SIV are CD4+ T cells in the gut. When these cells are destroyed, other CD4+ T cells rush to the gut to replace them, and there, they are also destroyed by the virus. The researchers hypothesised that stopping this cycle from being established could help bring the infection under control.
"We wanted to stop fuel being added to the fire, by stopping CD4+ T cells from migrating to the gut. On the surface of these cells, there is a molecule called alpha4/beta7 which enables them to lock to the gut. We hoped to target this molecule with anti-alpha4/beta7 to stop it from homing into gut-associated lymphoid tissues", explains Ansari.
Question that remains
Some interrogations remain and more research will needed to answer them. These include finding out how long the remission can last after the two year-period observed in the study and which parts of the immune system drive viral control.
Crucially, the scientists will need to find out if their results in monkeys can be replicated in humans and what the obstacles to translating them in clinical settings are. A pilot clinical trial is on its way, it has now began recruiting participants.
"The first order of business will be to see if the antibody has side effects in HIV positive patients, that we didn't see in monkeys. A similar antibody was FDA-approved for the treatment of Crohn's disease and ulcerative colitis in 2014 so we have a good indication that it will probably be safe but we never know. We hope to recruit 20 patients and finish this trial by next spring", concluded Ansari.
© Copyright IBTimes 2025. All rights reserved.