Immunotherapy drug combo to treat skin cancer approved for NHS use by Nice
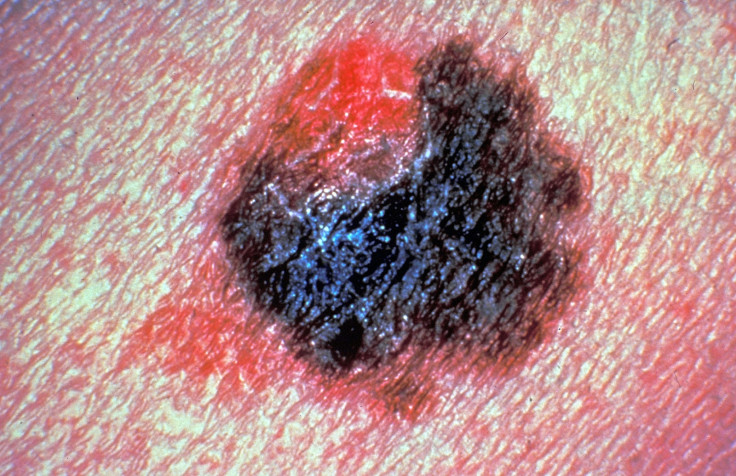
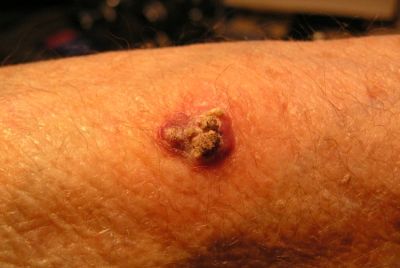
A combination of two immunotherapy drugs that treat advanced skin cancer has been approved for use on the NHS by The National Institute for Health and Care Excellence (Nice). The combination would benefit about 1,300 people diagnosed with advanced melanoma each year.
Following approval from the institute, the combination of Nivolumab and Ipilimumab will be used by the NHS to treat patients in England and Wales. The move comes after the combination was issued a licence a few months back.
It is backed by an ongoing study of 945 people diagnosed with advanced melanoma – a skin cancer variant that develops from the pigment-containing cells called melanocytes. Those administered the drug combination were found to live longer – an average 11.5 months – compared to patients taking only one of those drugs – like Nivolumab patients lived an average of 6.9 months longer, while patients taking Ipilimumab lived 2.9 months longer.
The findings were presented at a conference in Chicago, which was published in The New England Journal of medicine in July 2015.
Dr James Larkin, a melanoma specialist at the Royal Marsden, who worked on the study, was quoted by the Guardian as saying: "This is the first combination of immunotherapy drugs being approved by Nice. It is a milestone. It is fantastic to have this treatment option for patients."
Carole Longson, director of the health technology evaluation centre at Nice, added: "These promising new immunotherapy treatments for advanced melanoma look set to significantly extend the life of people with the condition. The evidence we examined was very promising and I know further trials are ongoing which have also released encouraging data."
© Copyright IBTimes 2025. All rights reserved.