Imperial College COVID-19 vaccine trial: First volunteer receives experimental inoculation
Once it has been administered, the vaccine then triggers an action wherein the body's cells attempt to recreate a copy of the virus' spike protein.
It was recently reported that the US government is funding six promising COVID-19 vaccine candidates for a hefty sum. As of now, among the biotech companies in the United States currently involved in clinical trials related to the pandemic, only Gilead Sciences and its remdesivir has been approved for emergency use.
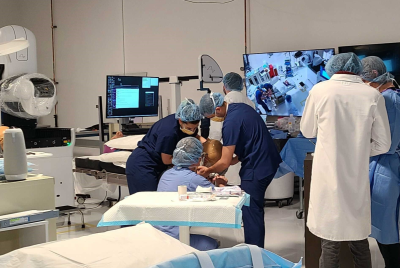
Meanwhile, in the United Kingdom, the Imperial College of London just confirmed that it has inoculated its first volunteer with its experimental SARS-CoV-2 vaccine.
The brave individual who preferred to remain unnamed but was revealed to have been given the dose at a West London medical facility. According to the team of researchers, the vaccine uses an innovative process called self-amplifying RNA (saRNA). This relies on advanced genetic engineering practices to tweak certain properties of ribonucleic acid that can potentially lead to even more groundbreaking discoveries down the line.
The way it works, as detailed by the experts, is to use the genetic code taken from a sample of the 2019 novel coronavirus. This allows them to craft a synthetic version that will be injected to a healthy patient. Once it has been administered, the vaccine then triggers an action wherein the body's cells attempt to recreate a copy of the virus' spike protein. What follows after is a natural response wherein the immune system attempts to create antibodies that will fight the infection.
The UK government is apparently funding the trials for approximately $51 million. The preliminary stages of testing see 15 volunteers submit to a low-dose treatment. The dosage will be increased for those that follow thereafter. Based on what was reported by other publications, many biotech companies are also using a similar approach.
Among other groundbreaking developments that were announced, another one from Oxford University highlighted the beneficial effects of a drug called dexamethasone. Results from a national study show that the drug was capable of reducing the risk of death among COVID-19 patients who experienced either moderate to severe symptoms. This will allow doctors to rely on these types of treatments for the meantime until a vaccine candidate is finally proven to be effective against the virus.
© Copyright IBTimes 2025. All rights reserved.