Parasitic worm genome sheds light on new treatment options for river blindness
Looking at the genome of worm Onchocerca volvulus may help scientists find new drugs to kill the parasite.

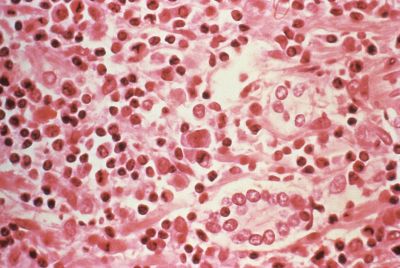
Scientists have for the first time fully sequenced the genome of onchocerca volvulus, the parasitic worm that causes river blindness. This has allowed them to identify a number of proteins that could be targeted by existing drugs to reduce the disease's incidence.
River blindness, also known as onchocerciasis, is a serious neglected tropical disease that is transmitted to humans when they are bitten by black flies of the genus Simulium who carry the parasite.
The World Health Organisation estimates that there are around 25 million people infected with O. volvulus globally, and 123 million people are at risk for becoming infected. The infections manifest clinically as debilitating skin disease and in some cases, vision impairment or blindness.
Although the disease has nearly disappeared from most of Latin America, where it was endemic, many cases still subsist, mostly in Africa.
"The entire control program of the disease relies on one drug known as ivermectin, which has eliminated onchocerciasis from all but two countries in the Americas", James Cotton, senior staff scientist at Wellcome Trust Sanger Institute who worked on the new research, told IBTimes UK.
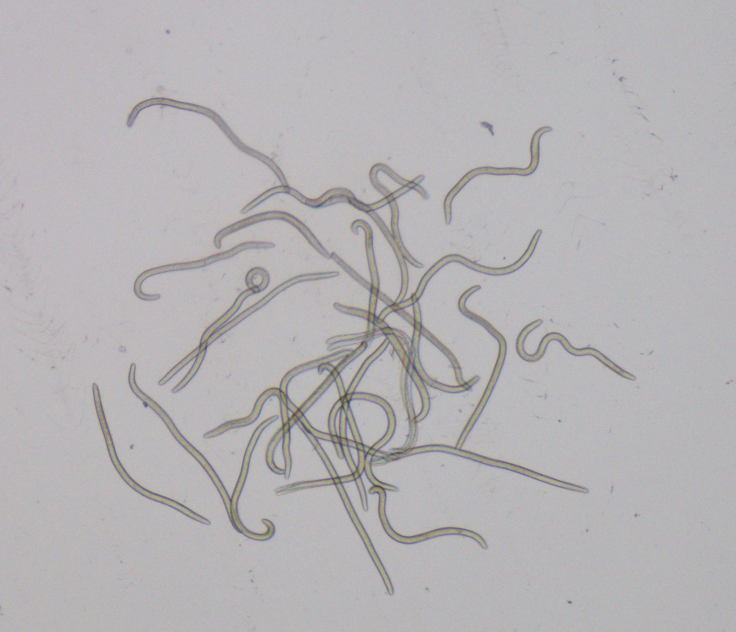
"The problem is that it only targets the larval form of the parasite and does not kill adult worms. Also, scientists working in Africa have started to see what could be the first signs of drug resistance".
Another problem is that O. volvulus can sometimes be found in the same areas as another parasite known as Loa loa. Ivermectin appears to cause serious adverse reactions in people who take it, when they are also severely infected by Loa loa, so in some regions of Central Africa, the drug cannot be given.
For all these reasons, the two latest studies on the subject published in Nature Microbiology look at the genome of O. volvulus, to come up with new therapeutic solutions.
Protein-coding genes and DNA variations
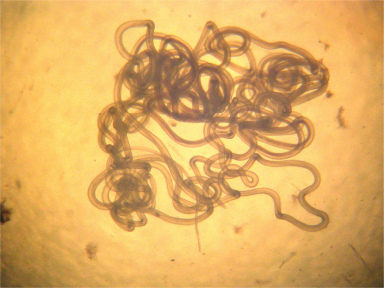
The first study, led by Sara Lustigman and to which Cotton participated, presents a full genomic sequencing of O volvulus, recovered from infected black flies. It identifies more than 12,000 protein-coding genes that are expressed across the eight stages of the parasite's life-cycle.
The authors suggest the genome and associated data sets will enable the development of new and urgently needed interventions against river blindness and other diseases associated with parasitic worm infection.
In the second study, Makedonka Mitreva and colleagues describe the genomic diversity of O. volvulus, showing there are important variations in the nuclear and mitochondrial DNA of the worm.
The findings of both research papers may help improve ongoing control programmes and could also help the scientists to identify existing drugs that can be repurposed to kill the worms and treat people.
In the first paper, the researchers in fact identify 51 O. volvulus proteins, mostly enzymes and proteins involved in ion transport and neurotransmission, which could be targeted by 85 drugs already approved by the US Food and Drugs Administration.
"We have also built was is known as a 'metabolic model' – a mathematical model that offers an insight into the chemical reactions that can occur in the worm – so that we can predict which reactions O. volvulus needs to survive, and can identify the genes that allow these reactions to occur. Some of our colleagues are now testing these predictions experimentally, hoping to identify drugs that can target these genes", Cotton concludes.
© Copyright IBTimes 2025. All rights reserved.