Malaria vaccine breakthrough following study on immune response to infection
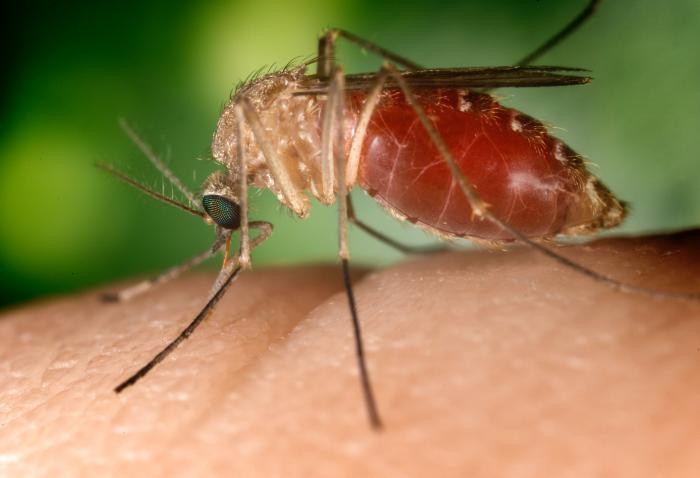
Melbourne researchers have made a significant discovery that can bring a viable malaria vaccine closer to reality by understanding how the human body's immune system fights the infection.
Although malaria is transmitted by mosquitoes, it becomes harmful when the infection is replicated in the body's red blood cells.
Professor James Beeson from the Burnet Institute and team set out to find what the immune system does or can do to block that and stop the infection.
They identified the body's immune system needs both antibodies and other proteins in the bloodstream to fight off malaria in what he called a "double-hit" effect.
Those antibodies recognise the parasites and then need to recruit other proteins that are in the bloodstream, known as complement proteins, to fight the infection.
"The two together, the antibodies and the complement, perform a double hit on the malaria infection and stop it from getting inside red blood cells, and therefore stopping the infection and the subsequent disease," said Beeson.
Experimental candidate vaccines have been seen to induce this antibody complement double-hit effect in some people, he said.
"So the step forward really is to say: how can we maximise that effect? How can we get high levels of antibodies and get the right type of antibodies that will recruit complement to maximise the protective effect of the vaccine?"
The work was published in the international journal Immunity.
Drug-resistant malaria is a cause of concern at present with resistance growing against the combination therapy based on artemisinin, a Chinese herb derivative.
Recent detection of artemisinin resistant strains of the pathogen at the Myanmar-India border raised alarms of a possible threat to millions across the globe if the resistance spreads across the border.
Deaths from mosquito-borne malaria have nearly halved since 2000, but the infection still killed about 584,000 people in 2013, mostly in Africa, according to the World Health Organization (WHO).
Much of the success was due to the use of combination therapies (ACTs) based on artemisinin.
Back in the last century, chloroquine saved hundreds of millions of lives, but resistance was discovered in 1957 around the border between Cambodia and Thailand. Resistance spread around the world and reached Africa 17 years later.
Chloroquine was then replaced by sulphadoxine-pyrimethamine (SP), but resistance to SP subsequently emerged in western Cambodia and again spread to Africa.
SP was replaced by artemisinin combination treatment, or ACT.
© Copyright IBTimes 2025. All rights reserved.




















