Electromagnetic brain stimulation could quell cocaine addiction in humans
An Italian physicist has treated over 300 patients using the non-invasive technique.
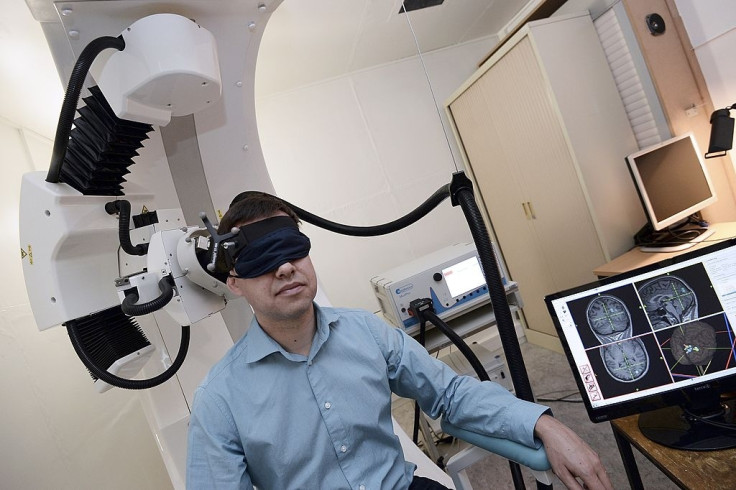
An Italian physicist, Luigi Gallimbert, has used electromagnetic brain stimulation to treat cocaine addicts successfully, according to a report in Futurism.
Gallimbert achieved this with the help of Transcranial magnetic stimulation (TMS) – a technique of using electromagnetic pulses to stimulate the brain's neural activity (firing or slowing neurons). He has managed to quell the urge to take cocaine in more than 300 addicts till now. The physicist used the non-invasive method to stimulate that particular area of the human brain which controls 'impulse' – a part of the prefrontal cortex sitting behind the forehead.
Gallimbert tested the technique on some 32 cocaine addicts under a pilot experiment. After noting promising preliminary findings, he started offering TMS-based treatment at $118 [£91] per session. Those who couldn't afford got the treatment for free.
Interestingly, the root idea for the treatment didn't come from Gallimbert. He borrowed the concept from a National Institute on Drug Abuse (NIDA) experiment conducted in 2013 by neuro scientists Antonello Bonci and Billy Chen to demonstrate the possibility of kicking cocaine addiction through magnetic stimulation. The only difference was that they tested the stimulation on rats, instead of humans.
The neurologists trained rats to pursue cocaine and modified them genetically to control their neural activity with light. Then, the same steps followed: they stimulated the area responsible for impulse control. It kicked the habit instantaneously in the animals that tolerated repeated electric shocks just to get their fixes. Bonci had posited the same could be done via TMS to alleviate drug addiction in humans.
Gallimbert and Bonci's work builds on the idea that TMS could soon be the answer to one of the deadliest addictions on the planet, affecting about 13 million people worldwide. The technique has already been approved by the FDA for treating depression and it is relatively inexpensive.
For now, several research institutes are working to test the efficacy as well as possible side-effects of TMS. NIDA will conduct a controlled trial for the method with 60 patients sometime next year.
© Copyright IBTimes 2025. All rights reserved.




















