Wales Becomes First British Country to Approve Cannabis-Based Medicine

Wales is to become the first country in the UK to approve a cannabis-based medicine for those suffering with multiple sclerosis.
The drug Sativex, a spray that is administered orally, has been approved by the All Wales Medicines Strategy Group (AWMSG) and Welsh health minister Mark Drakeford.
The drug will be given to MS patients experiencing muscle spasms and stiffness who have not had positive results from other medicines.
Sativex was blocked for use in England after the National Institute of Health and Care Excellence (NICE) decided not to use it in its draft clinical guideline for MS treatment.
However, following the approval by Drakeford and the AWMSG for its use, Wales will now be the only country in the UK where it will be available.
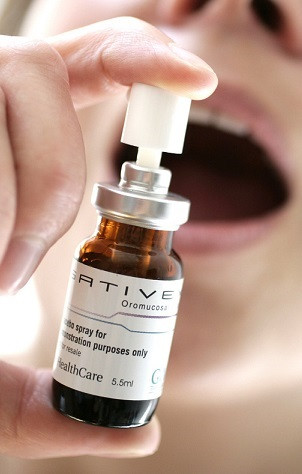
The move was described as a "significant milestone" for the treatment of conditions such as MS and spasticity.
Following the announcement of the first cannabis-based medicine available in the UK, Drakeford said: "I am pleased to announce we will be making the medicine available on the Welsh NHS to those who need it.
"I hope this decision will help ease the suffering of some of those who have to live with the reality of MS every day."
Sally Hughes, programme director for policy at the MS Society, said Wales is now "leading the way" in terms of MS treatment in the UK.
"Muscle spasms and stiffness in MS can be painful and distressing and so the availability of a treatment that can potentially alleviate these symptoms is good news," she said.
"We particularly welcome this decision considering the draft NICE clinical guideline, published in April, rejected this treatment for use on the NHS in Wales and England based on a flawed assessment of the drug's cost effectiveness.
"For some time we've been aware of people in Wales paying privately for this licensed treatment – this decision should make life a lot easier for them."
Amy Bowen, director of service development at the Multiple Sclerosis Trust, said: "We are extremely pleased that people with MS in Wales will finally have better access to Sativex.
"As a charity we have campaigned over a long period for Sativex to be widely available because of the significant impact that MS spasticity can have on daily activities.
"We just hope that this recommendation will now lead to Sativex being more easily accessible in the rest of the UK."
© Copyright IBTimes 2025. All rights reserved.





















