Periodic table creator Russian chemist Dmitri Mendeleev honoured by Google Doodle

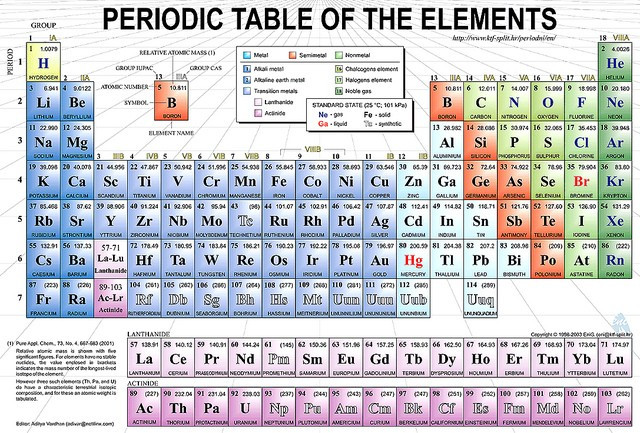
The father of the periodic table, Russian chemist Dmitri Mendeleev has been honoured by a Google Doodle to mark his 182nd birthday on 8 February. The table was published in 1869 and correctly organises the 63 known elements based on their atomic mass.
But most importantly, Mendeleev left gaps for missing elements, correctly predicting that these spaces would be filled by those yet to be discovered. He also predicted the properties of five elements that had yet to be discovered.
Three of them were discovered during his lifetime - gallium (1875), scandium (1879) and germanium (1886). The discovery gave further credence and acceptance of the periodic table among the scientific community. The other two elements whose properties were predicted were only discovered after 50 years.
An element, atomic number 101 was named after Mendeleev to honour him as the formulator of the Periodic Table, The radioactive element was named Mendelevium in 1955. So how did he discover the Periodic Table?
The Royal Society of Chemistry, said that Mendeleev had written the properties of elements on pieces of cards and tradition has it that after organising the cards, he suddenly realised that by arranging the element cards in order of increasing atomic weight that certain types of element regularly occurred.
For example, a reactive non-metal was directly followed by a very reactive light metal, then a less reactive light metal. Shortly after, his ideas were presented to the Russian Physico-chemical Society. They were read by Professor Menschutkin because Mendeleev was ill at the time. His ideas were then published in the main German chemistry periodical of the time, Zeitschrift fur Chemie.
Mendeleev also wrote about mixing alcohol and water, which apparently led to vodka being a standard 40%. The chemist, who was born in 1834 in Siberia, died of flu in St Petersburg at the age of 72. In addition to having an element named after him, he also has the honour of having a crater on the moon named after him.
The table has since been expanded and further refined over the years. Its seventh row was finally completed only last month. The International Union of Pure and Applied Chemistry announced synthetic elements - 113 (Ununtrium,) 115 (Ununpentium) , 117 (Ununseptium ) and 118 (Ununoctium) permanent places on the table on 5 January.
© Copyright IBTimes 2025. All rights reserved.


























